When a mouse team has a small cohort and limited serum, plasma, or tissue material, the "pilot" is rarely the smallest version of the full study.
More often, it's a pressure test: Is this matrix workable? Is there any measurable separation between groups? Is the signal stable enough to justify spending the rest of the samples?
That's why a 10-sample quote request can be a trap. If the pilot isn't scoped to answer a specific next-step decision, it can consume irreplaceable material and still leave the team uncertain about what to do next.
This guide is written for consideration-stage researchers planning an Olink mouse pilot study with real constraints—small N, small volume, and early-stage biomarker questions. It's especially relevant when the project looks like a small cohort Olink mouse study (e.g., N≈5 per group) and the team can't afford to spend samples twice. The goal is not to promise publication-grade inference from a tiny cohort. The goal is to help you design a pilot that produces decision-worthy information.
**Key Takeaways**:
A small pilot should de-risk one (or at most two) uncertainties—matrix feasibility, signal detectability, or scale-up readiness.
Start with a narrow pilot objective, keep matrices disciplined, spend your limited budget on biological coverage before duplicating everything, and define go/no-go criteria *before* you thaw the first tube.
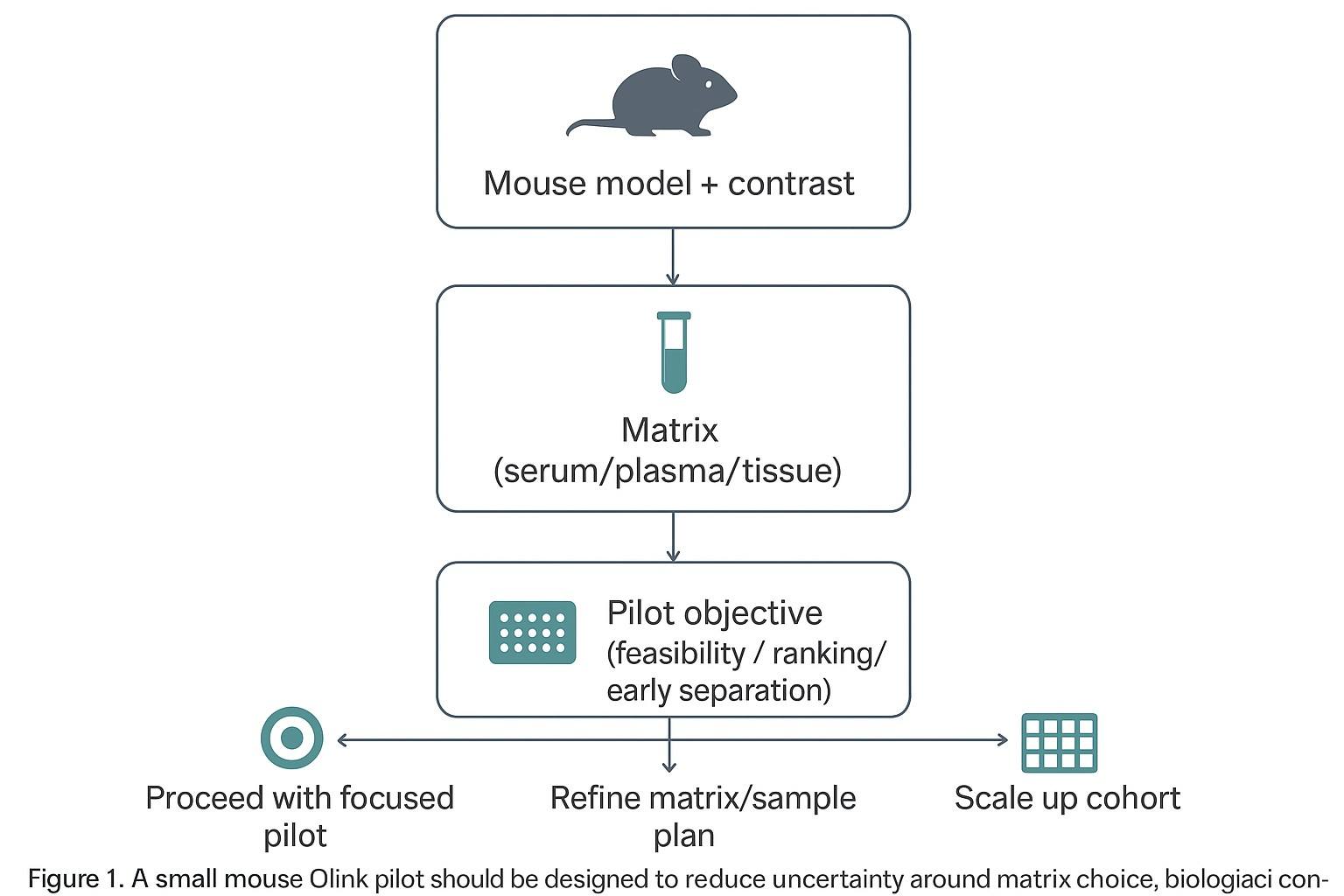
What a mouse Olink pilot should do—and what it should not try to do
A pilot is meant to de-risk the next step
A pilot is valuable when it reduces uncertainty about what you should do next. For small mouse cohorts, that usually means choosing among three practical outcomes:
- Proceed with the planned contrast (disease vs control, treated vs untreated) and scale up the cohort
- Refine the matrix or preanalytics plan before spending more samples
- Stop or pivot because the current design can't answer the question with the available material
The NC3Rs describes pilot studies as small experiments used to resolve logistics and gather information before a larger study—checking feasibility of procedures, measures, and adverse effects rather than trying to "win" a biological conclusion in miniature.
A pilot is not the same as a definitive biological conclusion
Small pilots are often underpowered for broad claims. Even in human feasibility research, pilot guidance emphasizes that small samples produce imprecise estimates and wide uncertainty intervals—making pilots unreliable for effect-size estimation and definitive inference (see feasibility pilot study guidelines (Teresi et al., 2022)).
Mouse biomarker pilots have an added constraint: your inference space is narrow. You're often evaluating one strain, one facility, one batch of animals, and a single set of collection conditions. Treat the pilot as an instrument for design decisions.
The biggest mistake is trying to answer too many questions at once
Most waste in small mouse pilots isn't caused by the assay. It's caused by scope creep:
- adding too many matrices "just in case"
- expanding the biomarker question into discovery + mechanism + validation
- duplicating everything without knowing whether variability is dominated by biology or handling
A disciplined pilot is intentionally incomplete. It is designed to justify the next spend.
Start with the biological question before choosing serum, plasma, tissue, or all three (mouse biomarker discovery pilot)
Serum and plasma pilots answer different practical questions than tissue pilots
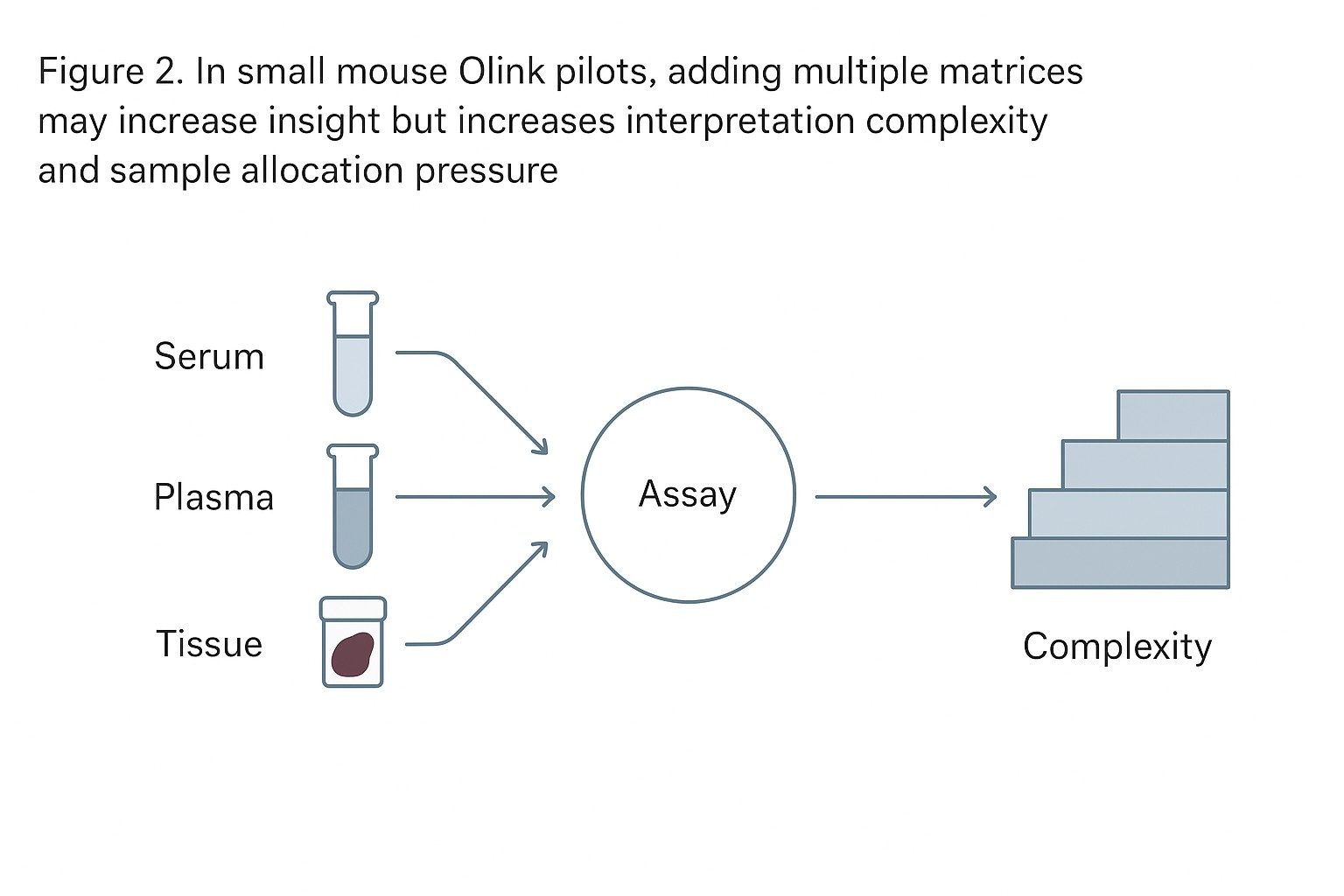
In small mouse cohorts, matrix is not a cosmetic choice. It changes what you can realistically learn.
- Serum/plasma pilots are usually the fastest route to assessing detectability and between-group separation for circulating proteins.
- Tissue pilots may be more proximal to the mechanism, but they can amplify heterogeneity (regional sampling, necrosis, perfusion differences, cell-type composition) and increase preanalytics burden.
If your decision is "Should we scale a circulating biomarker study?", a plasma/serum pilot that tests collection and handling consistency is often more decision-relevant than adding tissue by default.
Mixed-matrix pilots are tempting, but they can dilute interpretability
A mixed-matrix pilot (e.g., serum + tissue) feels efficient: one cohort, multiple answers. But with small N and scarce material, each additional matrix does three things:
- Consumes allocation bandwidth (you split your sample budget across matrices)
- Adds preanalytics variability (different collection, processing, stabilization, normalization assumptions)
- Creates interpretation branching (discordant results become harder to act on)
That's not an argument against tissue. It's an argument for deciding what you want the pilot to de-risk.
If your pilot includes multiple matrices, define why each one is there
A practical rule: every matrix must earn its seat by answering a different decision question.
For example:
- Plasma: "Can we detect a stable circulating signal that separates disease vs control?"
- Tissue: "Do we see directionally consistent biology in the affected organ that helps interpret plasma hits?"
If both matrices are included "because we have them," you're likely to burn samples without gaining clarity.
Table. Questions to ask before including more than one matrix in a mouse Olink pilot
| Matrix option | Why researchers include it | What it may clarify | What complexity it adds |
| Single matrix (plasma or serum) | Keep design simple; maximize n per group | Feasibility + signal detectability + early separation | Lowest complexity; fewer confounders |
| Single matrix (tissue) | Proximity to mechanism | Tissue-local changes; pathway relevance | Sampling heterogeneity; processing variability |
| Plasma/serum + tissue | Link circulating + local biology | Cross-matrix concordance; interpretability of hits | Sample allocation pressure; discordant results |
| Plasma + serum | Compare anticoagulant/processing impacts | Matrix effects for certain proteins | Double handling burden; hard to justify at small N |
How many animals are enough for an Olink mouse pilot study to be informative?
Small N can still be useful if the pilot question is narrow
A small cohort can be informative when you ask it a narrow question:
- "Is this matrix feasible with our collection workflow?"
- "Do we see any signal above background in this model?"
- "Are the top-ranked proteins stable enough to justify scale-up?"
Those are feasibility and prioritization problems, not definitive hypothesis testing.
A 10-animal pilot should not be expected to do the work of a full study
If you expect a 10-animal pilot to deliver clean p-values across dozens of proteins with strong generalizability, you're setting it up to fail.
Pilot feasibility guidance in other domains is explicit: small pilots produce unstable effect-size estimates and wide uncertainty, making them poor tools for powering the main study or drawing strong conclusions (see feasibility pilot study guidelines (Teresi et al., 2022)).
In practice, a 10-animal pilot is often best treated as:
- a matrix + workflow validation (preanalytics, handling, storage discipline)
- an early signal screen to inform whether scale-up is justified
Define whether your goal is feasibility, ranking, or early biological separation
Before you choose "N=5 per group," decide which of these you're trying to buy:
- Feasibility: Can you collect/process/store the matrix in a way that passes QC and yields analyzable data?
- Ranking: Can you prioritize a small set of proteins that look promising for follow-up?
- Early separation: Do disease vs control groups show any consistent directional separation worth scaling?
If you can't say which outcome you need, you'll struggle to interpret the pilot—regardless of the results.
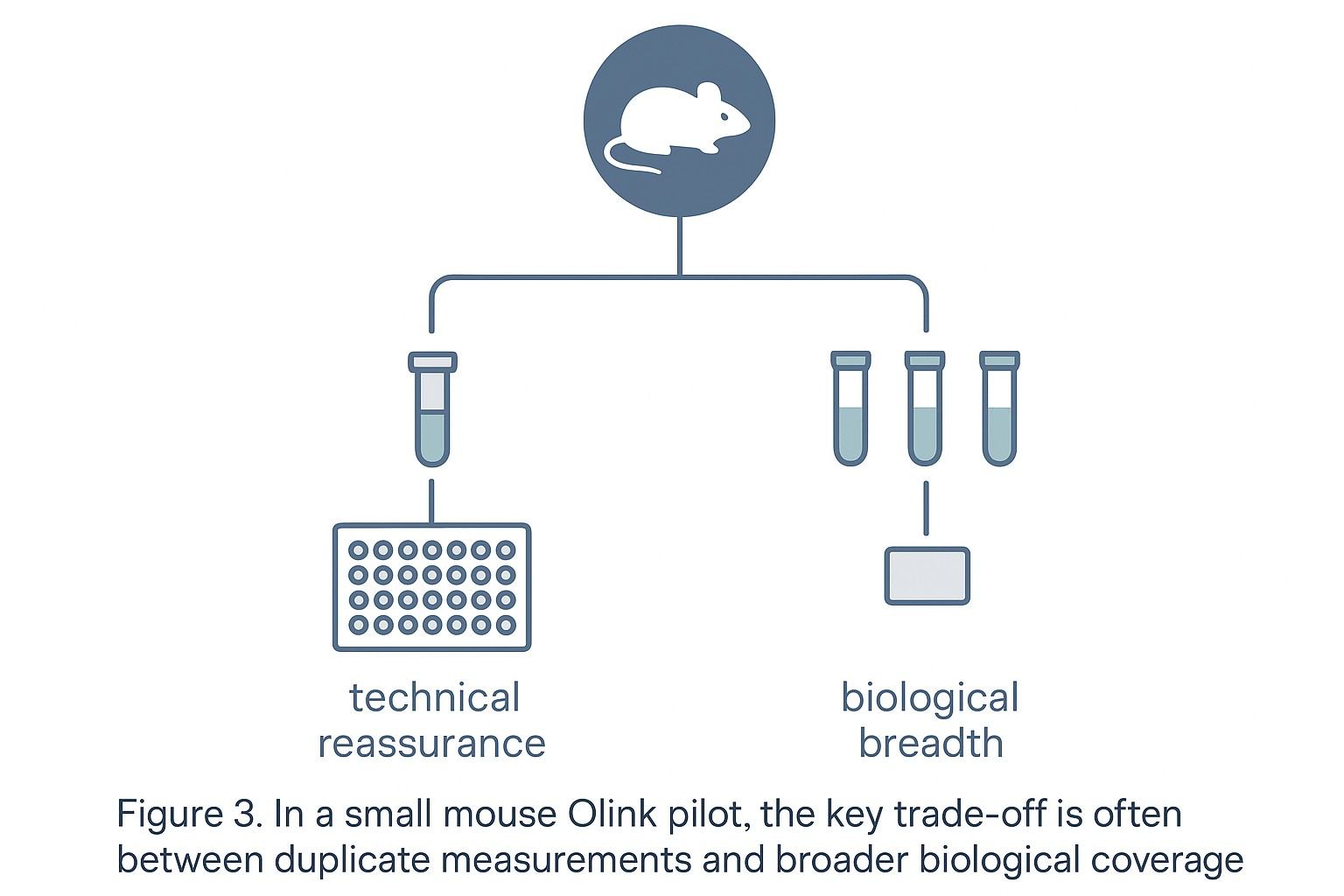
When to prioritize more biological animals instead of more duplicate measurements
Technical duplicates and biological animals answer different questions
- Technical duplicates test measurement repeatability on the same material.
- Biological animals test whether the signal generalizes across individuals and captures biological variability.
Both matter, but they buy different confidence.
For formal definitions and reporting expectations, refer to Nature's biological vs technical replicates guidance (PDF) and NIH/NIGMS training on biological vs technical replicates (PDF).
In small mouse pilots, extra animals often add more interpretive value
When your cohort is truly small, the limiting factor is usually biological inference. More independent animals typically improve your ability to:
- see whether apparent "hits" are stable or driven by one outlier
- understand within-group variability
- decide whether the model contrast is strong enough to scale
Duplicates can still be useful—especially if you have strong reasons to suspect preanalytics noise or plate/batch effects—but duplicating everything is often not the best use of a limited budget.
A subset-duplicate strategy may be smarter than duplicating everything
A practical compromise in small pilots is subset duplication:
- run duplicates for a small subset of samples (e.g., 10–25% of the cohort)
- prioritize duplication for samples most representative of the matrix (e.g., mid-volume, not hemolyzed, standard collection)
- use duplication to sanity-check variability and assay stability without halving biological coverage
If you need a deeper framework on this trade-off, see how to balance technical replicates vs sample limits.
Table. In a small mouse Olink pilot, where should limited sample budget go?
| Design choice | What it helps estimate | Main downside | Best use case |
| Add more animals | Biological variability; robustness of separation | May be constrained by model cost/ethics | You need a go/no-go decision for scale-up |
| Duplicate every sample | Technical repeatability | Cuts biological coverage; consumes material | You suspect technical instability dominates |
| Subset duplicates | Quick check of technical stability | Less precise than full duplication | You want some reassurance without burning N |
| Add matrices | Cross-matrix interpretation | Highest complexity + allocation pressure | Only if each matrix answers a distinct decision |
Mid-article CTA (medium intensity)
If you're scoping a small pilot and want a fast sanity-check before committing scarce material, share your mouse model, matrix, and pilot objective for a preliminary study-design discussion.
Which Olink formats are most sensible for small mouse pilots?
This section isn't a platform comparison. It's a scoping question: what breadth do you need to make the next decision? In other words: Olink pilot design mouse models should match the decision you need to make—not the maximum number of proteins you can measure. The same logic applies if your goal is an Olink mouse pilot without wasting samples.
When a focused mouse cytokine pilot makes the most sense
A focused panel is often the right pilot tool when:
- your hypothesis is immune/inflammation–driven
- your main uncertainty is matrix feasibility or early separation
- sample volume is tight and you want to preserve material for follow-up
For example, Olink's targeted mouse offering is designed for preclinical immune/inflammation work and is commonly positioned for low-volume input (see Olink Target 48 Mouse and Olink Target series panels).
When broader discovery formats become worth considering
Broader discovery formats become more defensible when:
- your pilot objective is explicitly mechanism discovery (not just feasibility)
- you have a realistic path to scale the cohort if the signal looks promising
- you can afford stronger controls and batch-aware design
Olink positions its ultra-high throughput discovery offering for large-scale discovery contexts (see Olink Explore HT and the broader Olink Explore series).
If you're planning something closer to a structured, control-heavy pilot at higher N, this reference can help: designing a larger Explore HT pilot with controls and go/no-go criteria.
Match panel breadth to the pilot decision—not to curiosity alone
A small pilot fails when it becomes a "discovery wish list" without a decision framework.
A simple scoping test:
- If the next decision is "Should we scale this contrast?" → prioritize decision clarity (often narrower).
- If the next decision is "Which pathways do we follow mechanistically?" → broader coverage may be justified, but only if you can scale and validate.
What should count as a successful pilot before you scale up?
Feasibility success is not the same as publication-ready biology
A pilot can be "successful" even if it doesn't produce statistically strong claims—if it clearly tells you what to do next.
The NC3Rs framing is useful here: pilots resolve feasibility and logistical risks so the main experiment is not built on a shaky foundation (see NC3Rs guidance on conducting a pilot study).
Define your go/no-go criteria before running the pilot
Define criteria that map to your pilot objective. Examples:
- Matrix pass/fail: sample quality and handling consistency meet your QC expectations
- Detectability: enough proteins are measurable above background to support ranking
- Stability signals: subset duplicates (if used) are directionally consistent
- Separation signal: group differences are directionally consistent across multiple animals (even if not "significant")
If you're running a targeted mouse panel, align your expectations with your downstream QC and deliverables requirements (see QC expectations for Olink Target 48 Mouse cytokine studies and audit-ready QC and deliverables for Target 48 Mouse projects).
Decide in advance what would justify expanding the cohort
Be explicit about what "scale-up" means:
- doubling animals per group?
- adding time points?
- narrowing to a short list of proteins for verification?
And decide what would stop the program. A pre-defined stop condition is part of sample preservation.
How to describe a small mouse Olink pilot clearly when requesting support
Teams often want a quote before the pilot logic is finalized. You can still get useful guidance—if you describe the problem cleanly.
State the model and the biological contrast first
Start with:
- species/strain
- disease vs control or treatment groups
- timing and expected phenotype window
Then define matrix, sample count, and whether material is limited
Be explicit about constraints:
- serum/plasma/tissue
- volume per sample (if known)
- whether samples are irreplaceable (neonatal, rare strain, terminal collection)
Finally explain what the pilot is supposed to de-risk
Use one sentence:
- "This pilot is to validate matrix feasibility and decide whether to scale the cohort."
- "This pilot is to rank candidate biomarkers for a follow-up confirmation study."
Before submitting your inquiry, clarify:
- species and model
- disease/control or treatment groups
- matrix type
- number of animals per group
- whether material is limited or irreplaceable
- whether duplicate measurements are being considered
- whether the pilot is for feasibility, ranking, or scale-up
For a structured intake format, see the Olink quotation checklist.
Common mistakes in small mouse Olink pilot design
Trying to answer discovery, validation, and mechanism at the same time
If your pilot tries to do everything, it often does nothing well. Decide the next decision you need to make—and design to that.
Mixing too many matrices in a small cohort
Mixed-matrix pilots can be high-value, but only when each matrix answers a distinct decision question. Otherwise, you buy complexity and lose clarity.
Confusing pilot feasibility with strong biological inference
Small pilots can validate feasibility and produce prioritization signals. They rarely justify strong biological claims without follow-up.
Spending scarce material before defining go/no-go logic
If you define success after you see the data, you risk rationalizing a weak pilot into a bigger spend.
**⚠️ Warning**: Even in a pilot, basic design hygiene matters. Randomization and blinding reduce bias, and controlling biological/environmental variables improves reproducibility (see [JAX reproducible mouse research design strategies](https://www.jax.org/news-and-insights/jax-blog/2016/march/experimental-design-top-four-strategies-for-reproducible-mouse-research)).
FAQ
1) How many mice are enough for an Olink mouse pilot study?
Enough depends on the decision you need to make. For feasibility and workflow validation, very small cohorts can be useful. For strong biological inference, small pilots are often underpowered and yield uncertain estimates (see feasibility pilot study guidelines (Teresi et al., 2022)).
2) Should I use serum, plasma, tissue, or more than one matrix in a small pilot?
Choose the matrix that best matches your next decision. Add a second matrix only when it answers a different decision question—and you can afford the sample allocation and interpretation complexity.
3) Is it better to add duplicate measurements or more animals?
In many small mouse pilots, more biological animals add more decision value because they better reflect biological variability. Duplicates add technical reassurance but can reduce biological coverage. A subset-duplicate strategy is often a workable compromise.
For definitions and reporting expectations, see Nature's biological vs technical replicates guidance (PDF) and NIH/NIGMS training on biological vs technical replicates (PDF).
4) Can a 10-sample pilot still be worth doing?
Yes—if the pilot objective is narrow (matrix feasibility, detectability, ranking). It's usually not enough to support broad biological claims across many proteins.
5) When should I choose a focused mouse cytokine panel instead of broader discovery?
Choose focused when your hypothesis is narrow (e.g., inflammation), sample volume is limited, and your next decision is scale-up readiness. Consider broader discovery when your objective is mechanism discovery and you have a realistic path to scale and validate.
See Olink Target 48 Mouse and Olink Explore HT.
6) What should count as a successful mouse pilot before scale-up?
Success should be defined as a decision outcome, not a p-value. Define go/no-go criteria before the run, aligned to feasibility (QC), detectability, and directionality of separation.
7) What information should I provide when requesting support for a mouse pilot?
Model, contrast, matrix, sample counts, constraints on volume/material, whether duplicates are planned, and what the pilot is meant to de-risk. Use the Olink quotation checklist as a template.
8) Can the same pilot support both feasibility and biomarker prioritization?
Often yes, if you keep the scope tight: validate feasibility first, then use the same data to rank a short list of candidates—with the understanding that prioritization is not final validation.
Conclusion
A good small mouse pilot is not the smallest version of a full study. It's a deliberately scoped preclinical proteomics pilot design that reduces uncertainty, preserves scarce material, and justifies the next step.
If you can clearly state your model contrast, matrix strategy, and pilot objective—and define go/no-go criteria before you run—you dramatically increase the odds that the pilot answers the question you actually care about: should we scale, refine, or stop without wasting samples?
Next steps (end CTA)
If you're planning an
(or plasma/tissue) pilot and need to keep it sample-efficient, share:
- model and group structure
- matrix (serum/plasma/tissue)
- number of animals per group
- whether material is limited or irreplaceable
- whether duplicate measurements are being considered
- whether the pilot is meant to guide a larger follow-up study





